- Home
- Publications
- PAGES Magazine
- Analytical Challenges and Advancements In Measuring Individual Mineral Nanoparticles and Microparticles Entrapped In Ice Cores
Analytical challenges and advancements in measuring individual mineral nanoparticles and microparticles entrapped in ice cores
Madeleine Lomax-Vogt, S. Kutuzov, P. Gabrielli and J. W. Olesik
Past Global Changes Magazine
31(2)
90-91
2023
Madeleine Lomax-Vogt1,2,
S. Kutuzov1,3,
P. Gabrielli4 and
J.W. Olesik1,3
Ice cores provide a record of atmospheric particles that could have affected past climate. Single-particle inductively coupled plasma time-of-flight mass spectrometry can measure size distributions, number concentrations, and elemental chemical composition of many thousands of individual insoluble nanoparticles and microparticles.
Particles in Earth’s atmosphere and climate
Atmospheric particles include sulfates, nitrates, black/organic carbon, sea salt, and mineral or other water-insoluble particles with diverse natural and anthropogenic sources (e.g. volcanic eruptions, desert dust, biomass burning, fossil-fuel combustion, sea spray). Compared to other climate forcings like carbon dioxide (CO2) and methane, atmospheric particles are irregularly distributed in the atmosphere, reflecting diverse sources, short lifetimes, and complex atmospheric transport conditions (Raes et al. 2000).
The chemical (e.g. mineralogy/elemental composition) and physical (e.g. size, solubility, density) properties of individual mineral atmospheric particles affect climate directly through absorption and scattering of solar radiation, and indirectly through changes in cloud and ice formation and structure (Haywood and Boucher 2000). Bioavailable iron-bearing particles can fertilize the ocean, creating algal blooms, thus, reducing the global CO2 concentration (Martin 1990). Particles with diameters <2.5 µm (“fine particulates”) containing toxic metals have negative impacts on human health (Shiraiwa et al. 2017).
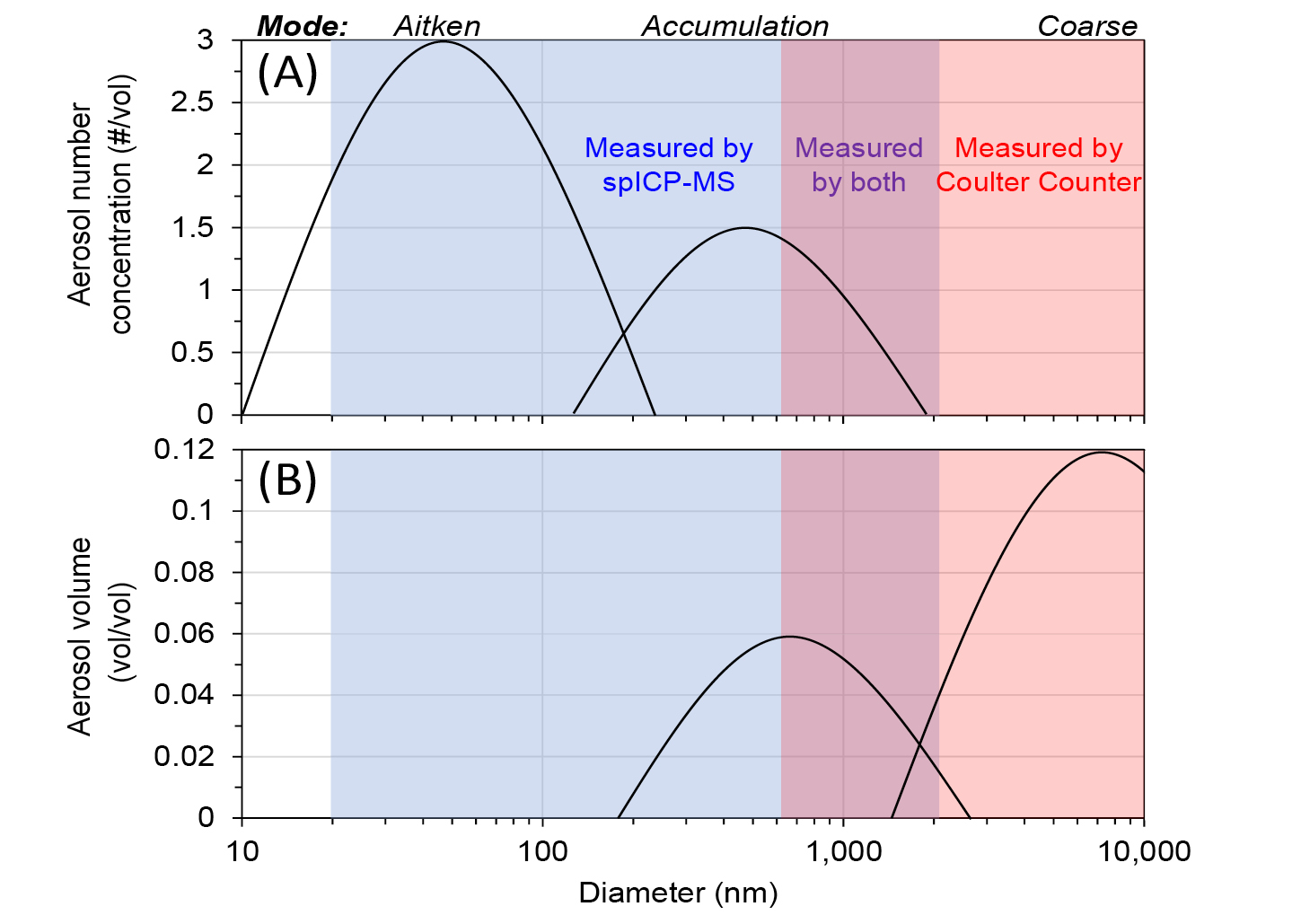
Most particles in the atmosphere and entrapped in ice cores (by number concentration) are classified within the Aitken mode size range (10 to 100 nm, nanoparticles), with a smaller but significant number within the accumulation mode size range (100 to 1000 nm, fine microparticles) (Seinfeld and Pandis 2006; see conceptual model particle size distributions in Fig. 1). However, particles within the Aitken mode contribute very little to the total mass (and volume) of particles, while most of the total mass (and volume) is in coarser particles. Glaciers are excellent archives of the Earth’s climate over hundreds of thousands of years, including deposited insoluble mineral atmospheric nanoparticles and microparticles. The physical and chemical characteristics of particles are indicative of the past atmosphere and can offer insight into how the Earth’s climate changed over time. Measuring particles entrapped in remote Antarctic ice can also provide a benchmark of comparison to the modern, anthropogenically affected atmosphere.
Here, we first discuss analytical techniques traditionally used to measure the particles entrapped in glacial ice cores. While useful, these methods cannot measure the elemental chemical composition of thousands of individual nanoparticles and fine microparticles. We then highlight single-particle inductively coupled plasma time-of-flight mass spectrometry (spICP-TOFMS) as a tool for measuring hundreds of thousands of individual mineral nanoparticles and microparticles.
Traditional techniques used to measure mineral atmospheric particles entrapped in glacial ice
Several analytical techniques, including Coulter Counter and Abakus, bulk analysis by inductively coupled plasma-sector field-mass spectrometry (ICP-SFMS), and transmission or scanning electron microscopy (TEM or SEM) with energy dispersive X-ray spectrometry (EDXS), are used to measure atmospheric particles in melted glacial ice.
Coulter Counters and Abakus are widely used to measure size distributions and number concentrations of accumulation and coarse mode insoluble particles larger than 500 nm (Fig. 1; Delmonte et al. 2002; Folden Simonsen et al. 2018) independent of the elemental-chemical composition of particles.
Bulk analysis by ICP-SFMS determines average elemental concentrations in an acidified sample (McConnell et al. 2018; Uglietti et al. 2014). It is impossible to differentiate between contributions from chemically digested insoluble mineral particles, water-soluble particles, and dissolved ions in the sample. Bulk analysis cannot measure particle-number concentrations, size distributions, or individual particle compositions. Measuring low-abundance trace elements (e.g. Ir, Pt) is challenging and requires ultra-clean techniques in order to avoid contamination (Gabrielli et al. 2004).
SEM/TEM-EDXS determines the shape, size, and elemental chemical composition/mineralogy of individual particles. SEM/TEM-EDXS is a time-consuming technique which requires locating and scanning each individual particle. The time required to measure a statistically significant population (i.e. thousands or hundreds of thousands) of particles in a sample is prohibitively long. A limited number of studies have used TEM/SEM-EDXS to measure mineral nanoparticles and microparticles, with most focusing on common particles containing elements present at major or minor abundances in the Earth’s crust (Ellis et al. 2015).
Techniques to measure elemental composition and size of thousands of individual particles
Single particle mass spectrometry (SPMS) has been used since the 1990s to measure particles in the modern atmosphere in real-time (Noble and Prather 2000). In the only reported use of SPMS to measure particles in ice cores (Osman et al. 2017), long sampling times (>1 hour) and more than 100 mL of liquid sample were required, which can be a limitation in analyzing ice cores with small sample volume. Furthermore, SPMS typically does not quantify the amount of each element in a particle. Instead, the amount of each element is reported as a percentage of the total ion current detected.
Single-particle inductively coupled plasma-mass spectrometry (spICP-MS) is becoming widely used to measure engineered nanoparticles in the environment, biological samples, and food (Montaño et al. 2016). The particle suspension is nebulized and introduced into the ICP ion source. Unlike bulk ICP-MS which measures the average concentration of elements over a few seconds, spICP-MS measures the ion signal with a time resolution as short as 10 µs. While spICP-MS using a quadrupole or sector field MS can only measure one element per particle, spICP-TOFMS measures a complete elemental mass spectrum (up to 70 elements excluding O, H, N, F, and the noble gases) every 30 µs. Each particle produces a discrete burst of signal ~500 µs long (“particle event”); instrument software identifies mass spectra due to a particle event. The relationship between ion-signal intensity and the mass of each detected element is calibrated using solutions so that the amount (femtograms -fg- =10-15 grams) of each detected element can be quantitated. From the amounts of each detected element, the percentage of each element in the particle can be estimated. There was only one previous publication (Erhardt et al. 2019) that used spICP-TOFMS to measure particles in an ice core to our knowledge.
Measuring insoluble mineral particles by spICP-TOFMS
spICP-TOFMS is uniquely capable of quickly (in ~30 minutes) measuring the estimated mass equivalent size distribution, number concentration, and elemental chemical composition of more than 100,000 individual insoluble mineral nanoparticles and microparticles using only one milliliter of melted ice.
The mass-equivalent particle diameter is estimated for each particle using the total mass (in femtograms) of all detected element(s) and an estimated density (i.e. average crustal density, 2.7 g cm-3). The shape of the particle cannot be determined by spICP-TOFMS. spICP-TOFMS can detect particles as small as ~20 nm (that are too small to be measured by a Coulter Counter; Fig. 1).
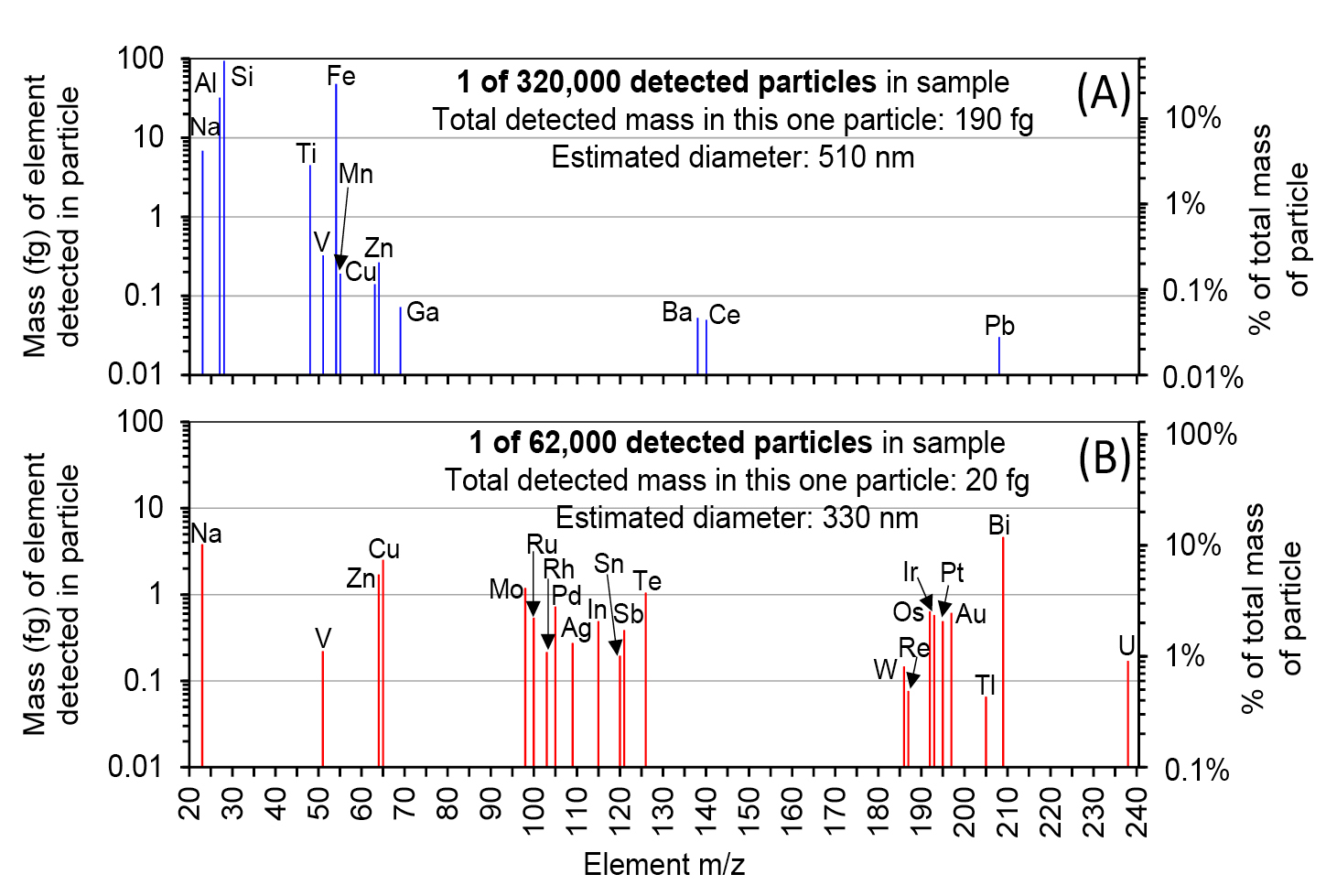
Figure 2 shows examples of mass spectra produced from two individual insoluble-mineral particles detected in Taylor Glacier ice samples by spICP-TOFMS. The top spectrum is from a common particle composed mainly of elements (e.g. Na, Al, Si, Fe, Zn, etc.) at major or minor concentrations in the Earth’s crust (Fig. 2a). The bottom particle contains ultra-low amounts (<10 fg) of uncommon elements such as Os, Au, Pt, Ir, and Tl (Fig. 2b). It would be difficult to measure these particles by bulk ICP-MS because the signal each element produces would be a very small fraction of the background signal integrated over several seconds.
Potential minerals and mineral groups can be inferred for each particle by comparing the ratio of the amounts of detected elements to known mineral elemental ratios. Common minerals (e.g. quartz, ilmenite, kaolinite) can be differentiated from particles with unusual compositions that would be undetectable by bulk ICP-SFMS, or challenging to find by SEM/TEM-EDXS. These particles can act as markers of very rare environmental events (i.e. micrometeorites; volcanic tephra from eruptions).
Conclusions and perspectives
Glacial ice cores contain crucial information about the size, number concentration, and elemental chemical composition of hundreds of thousands of insoluble mineral particles over Earth’s climatic and environmental history that were unmeasurable using conventional techniques. Measuring additional ice-core samples from the glacial–interglacial transition by spICP-TOFMS will give us a better understanding of the impact of mineral particles on past climate, and answer questions about how particle mineralogy, size, and number concentration changed over the glacial–interglacial timescale.
affiliationS
1Trace Element Research Laboratory, School of Earth Sciences, The Ohio State University, Columbus, USA
2Department of Chemistry and Biochemistry, The Ohio State University, Columbus, USA
3Byrd Polar and Climate Research Center, The Ohio State University, Columbus, USA
4Honeybee Robotics, Altadena, USA
contact
John Olesik: Olesik.2@osu.edu
references
Delmonte B et al. (2002) Clim Dyn 18: 647-660
Ellis A et al. (2015) Atmospheric Meas Tech 8: 3959-3969
Erhardt T et al. (2019) Environ Sci Technol 53: 13275-13283
Folden Simonsen M et al. (2018) Clim Past 14: 601-608
Gabrielli P et al. (2004) J Anal At Spectrom 19: 831-837
Haywood J, Boucher O (2000) Rev Geophys 38: 513-543
Martin JH (1990) Paleoceanography 5: 1-13
McConnell JR et al. (2018) Proc Natl Acad Sci 115: 5726-5731
Montaño MD et al. (2016) Anal Bioanal Chem 408: 5053-5074
Noble CA, Prather KA (2000) Mass Spectrom Rev 19: 248-274
Osman M et al. (2017) Atmospheric Meas Tech 10: 4459-4477
Raes F et al. (2000) Atmos Environ 34: 4215-4240
Shiraiwa M et al. (2017) Environ Sci Technol 51: 13545-13567