- Home
- Publications
- PAGES Magazine
- Clathrate Hydrates of Air In Polar Ice and Their Importance For Climate Science
Clathrate hydrates of air in polar ice and their importance for climate science
Florian Painer, S. Kipfstuhl and I. Weikusat
Past Global Changes Magazine
31(2)
82-83
2023
Ancient air bubbles trapped inside polar ice sheets transform into clathrate hydrates at a certain depth. Not only do they contain the gas molecules used for paleoclimatic reconstructions, but they also serve as a climate proxy themselves.
Clathrate hydrates are solid guest-host compounds, formed by small molecules (guests; e.g. N2, O2, CH4 or CO2) trapped in a crystalline framework (host) of hydrogen-bonded water molecules (Chazallon and Kuhs 2002). In natural environments, clathrate hydrates occur in deep-sea sediments and permafrost (e.g. as methane hydrates). The first direct observations of clathrate hydrates of air (hereinafter, air hydrates) in polar ice were made in 1982 by Shoji and Langway (1982) in the Dye-3 ice core (Greenland) using an optical microscope. Subsequently, air hydrates have been found in all deep ice cores in Greenland and Antarctica (Uchida et al. 2014).
Air hydrate formation in polar ice
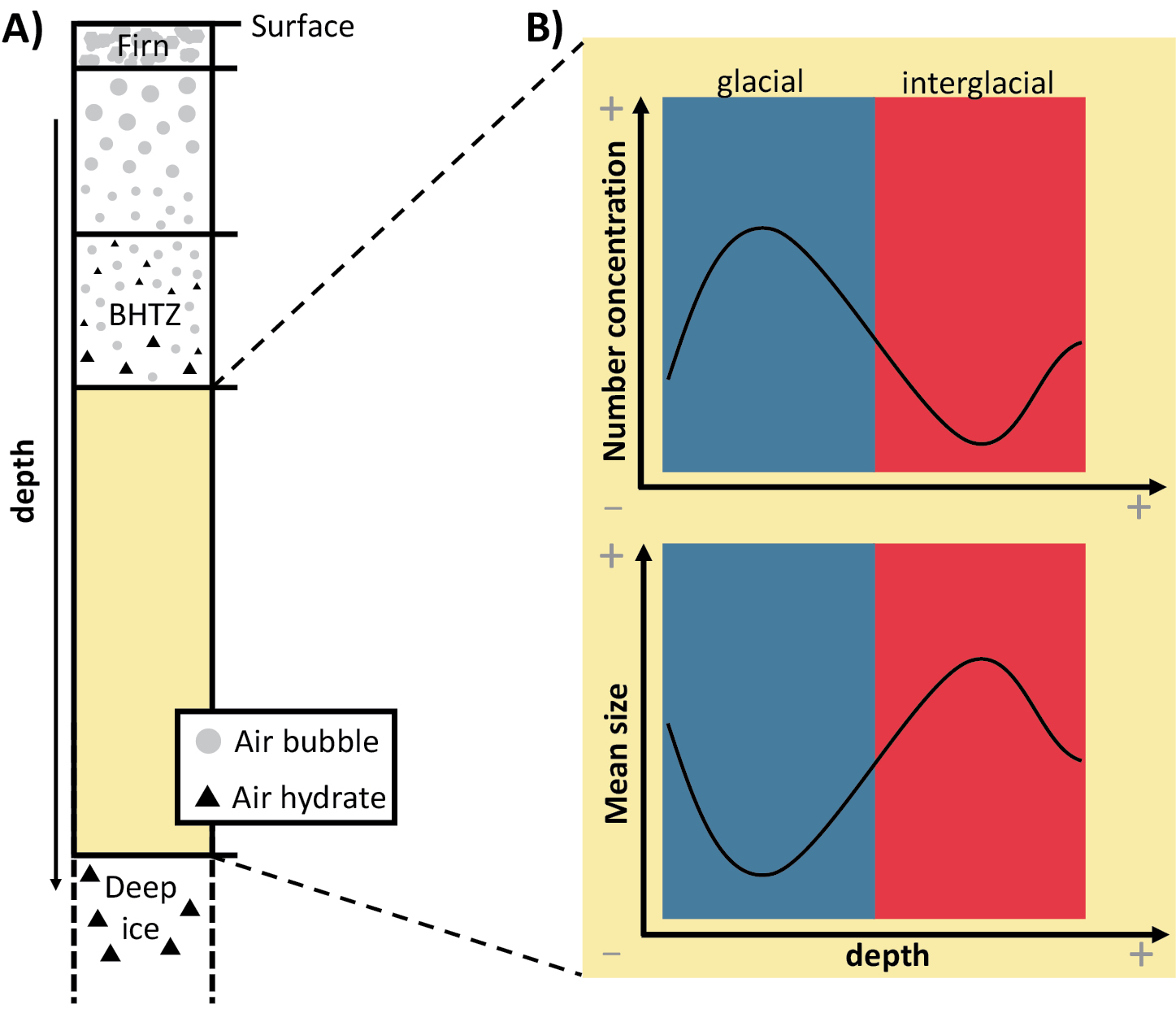
Air bubble inclusions in polar ice sheets are compressed with depth due to the increasing overburden pressure (e.g. Shoji and Langway 1982; Uchida et al. 2014). They transform into air hydrates below a certain depth, where the hydrostatic pressure enters the stability range of the clathrate hydrate at the in situ temperature (Uchida et al. 2011). The conversion occurs over a certain depth range, called the bubble-hydrate transition zone (BHTZ) (Fig. 1a), where bubbles and air hydrates coexist (Uchida et al. 2014). Its upper boundary is determined by the first appearance of a hydrate, the lower boundary by the last appearance of an air bubble.
For air-hydrate crystallization in the BHTZ, their (heterogeneous) nucleation is considered to be the rate-limiting step (Lipenkov 2000). Therefore, the starting depth and extent of the BHTZ varies (several hundred meters) for different ice-core sites in Greenland and Antarctica, owing to site-specific temperature and pressure conditions, as well as the occurrence of nucleation promoting or inhibiting factors (Uchida et al. 2014).
Given that air hydrates are formed from air bubbles, one could conclude that one bubble converts to one hydrate. However, a detailed investigation of the NGRIP (Kipfstuhl et al. 2001) and the Dome Fuji (Ohno et al. 2004) ice cores revealed that the number of total air inclusions (bubbles + hydrates) in the upper part of the BHTZ exceeds the air-bubble concentration above the transition zone.
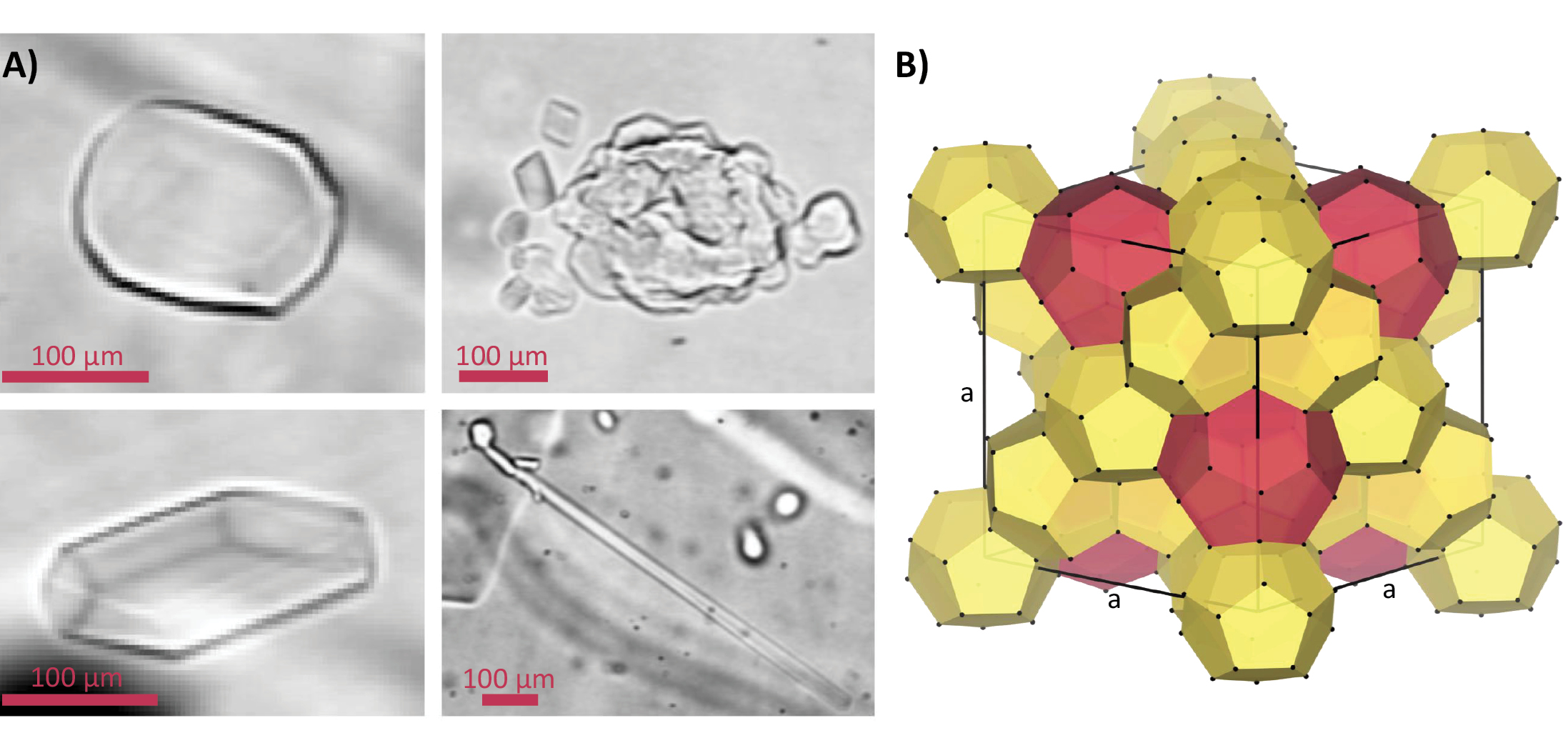
Moreover, air hydrates in the transition zone show a wide variety of morphologies (Fig. 2a) and a non-uniform spatial distribution compared to air bubbles (Kipfstuhl et al. 2001; Lipenkov 2000; Ohno et al. 2004). The interpretation is that their formation is more complex (e.g. involving impurities; Ohno et al. 2004) than a simple one-to-one conversion and needs to be reinvestigated, including post-formation splintering and recrystallization mechanisms (Kipfstuhl et al. 2001).
Air hydrates and paleoclimate
Air hydrates are the source of at least two different types of information on paleoclimatic conditions. Firstly, they contain most of the air molecules in ice below the BHTZ and, as a result, provide a unique archive to reconstruct changes in past atmospheric gas compositions (Bereiter et al. 2015; Lipenkov 2000; Uchida et al. 2011, 2014). Secondly, some of the air hydrates’ geometrical characteristics (mean size and number concentration) correlate with past climatic changes (Lipenkov 2000). This has been shown for the Vostok and Dome Fuji ice core in Antarctica and the GRIP ice core in Greenland, where continuous records of air hydrates exist (Ohno et al. 2004).
In general, ice formed at glacial conditions is characterized by smaller air hydrates and a higher hydrate number concentration compared to ice formed at interglacial conditions (Fig. 1b) (Lipenkov 2000; Salamatin et al. 2003). This is essentially explained by the impact of past temperature and accumulation rate on the physical properties of the firn. For example, smaller ice-crystal grain sizes in colder firn lead to smaller air bubbles in ice. The original climate-dependent geometrical properties of air bubbles are then transferred to air hydrates (Lipenkov 2000).
After their formation, air hydrates are subject to an evolution with depth: the general trend of number concentration continuously decreases, coupled with an increase in the average size (Uchida et al. 2011). This phenomenon can be caused by a displacement of the hydrates and further fusion, and/or the diffusion of gas molecules and the subsequent growth of larger air-hydrate crystals at the expense of smaller hydrate crystals (a process called Ostwald ripening) (Salamatin et al. 2003; Uchida et al. 2011). This is especially pronounced in the deeper parts of the ice sheet, where it can even alter the original, climate-dependent variation of geometrical properties (Salamatin et al. 2003; Uchida et al. 2011). Increasing depth (and pressure), changes in temperature, and increasing age of the ice are the fundamental parameters behind this phenomenon (Uchida et al. 2011).
Material properties and analytical methods for air-hydrate studies
X-ray diffraction studies on single crystals of natural air hydrates in the Dye-3 ice core revealed the Stackelberg’s crystallographic structure II (sII) (Hondoh et al. 1990). It is one of the three main structures found for natural clathrate hydrates. The sII has a cubic unit cell which consists of 16 small cages and eight large cages formed by 136 water molecules (Fig. 2b) (Chazallon and Kuhs 2002). One or two guest molecules can be enclosed in each polyhedral cage, but not all cages need to be occupied. The nature of the guests, temperature, and pressure determine the crystal structure of the host framework and the degree of filling of the cages (Chazallon and Kuhs 2002).
Studying air hydrates in polar ice is challenging because they are thermodynamically unstable under temperature and pressure conditions in the field, or in the cold-laboratories, where samples are processed and inspected. However, the surrounding ice acts as a pressure cell to keep the hydrates metastable for a certain time, which depends on the ambient temperature conditions (Uchida et al. 1994). Consequently, for the long-term preservation of the record, appropriate storage conditions for the ice cores (≤ -50°C) are necessary (e.g. Bereiter et al. 2015; Uchida et al. 1994).
Optical light microscopy and cryo-Raman spectroscopy are the main non-destructive methods used to analyze air hydrates in polar ice. The microscope is the go-to tool for studying air-hydrate geometric properties. This investigation needs to be made as soon as possible after core retrieval, especially for the study of hydrates of the BHTZ (Kipfstuhl et al. 2001). Cryo-Raman spectroscopy enables the study of the main air-hydrate guest molecules (nitrogen and oxygen). In addition, this method can be used to analyze the various air-hydrate morphologies in 3D (Weikusat et al. 2015).
Outlook
There are still many open questions regarding the formation and evolution of air hydrates in polar ice. In the framework of Beyond EPICA and other Oldest Ice projects, a high analytical resolution and the development of new methodologies and climate proxies (e.g. dating via 81Kr decay or 40Ar/38Ar isotope ratio in entrapped air; Lipenkov et al. 2019) play a key role in analyzing highly thinned, and likely disturbed, deepest and oldest ice in Antarctica. A better understanding of the air-hydrate formation processes and their physicochemical properties is essential for using entrapped air in ice cores as a climate proxy. Certainly, this will improve the evaluation of measured gas concentrations (e.g. O2/N2 ratios or CO2; Bereiter et al. 2015). Equally important are the processes related to their evolution with depth (age) to further develop dating of very old (and disturbed) ice (Lipenkov et al. 2019).
affiliationS
1Alfred Wegener Institute, Helmholtz Centre for Polar and Marine Research, Bremerhaven, Germany
2Department of Geosciences, Eberhard Karls University, Tübingen, Germany
contact
Florian Painer: florian.painer@awi.de
references
Bereiter B et al. (2015) Geophys Res Lett 42: 542-549
Chazallon B, Kuhs WF (2002) J Chem Phys 117: 308-320
Hondoh T et al. (1990) J Incl Phenom Molec Recog 8: 17-24
Kipfstuhl S (2007) Thick-section images of the EPICA-Dronning-Maud-Land (EDML) ice core. PANGAEA
Kipfstuhl S et al. (2001) Geophys Res Lett 28: 591-594
Lipenkov VY et al. (2019) Geophys Res Abstracts 21: EGU2019-8505
Momma K, Izumi F (2011) J Appl Cryst 44: 1272-1276
Ohno H et al. (2004) Geophys Res Lett 31: 1-4
Salamatin AN et al. (2003) J Cryst Growth 257: 412-426
Shoji H, Langway CC (1982) Nature 298: 548-550
Uchida T et al. (1994) Mem Natl Inst Polar Res 49: 306-313
Uchida T et al. (2011) J Glaciol 57: 1017-1026
Uchida T et al. (2014) J Glaciol 60: 1111-1116