- Home
- Publications
- PAGES Magazine
- In Situ Production of N2O In Ice Challenges Past N2O Reconstructions
In situ production of N2O in ice challenges past N2O reconstructions
Lison Soussaintjean, J. Schmitt and H. Fischer
Past Global Changes Magazine
31(2)
80-81
2023
The production of N2O in glacial ice alters the record of past atmospheric concentrations of N2O in ice cores. Using isotope analyses of N2O would help understand the production processes and, thus, isolate the atmospheric signal.
Air bubbles trapped in ice cores represent the only direct paleo-atmospheric archive, and allow for, for example, the reconstruction of past greenhouse gas concentrations. But ice cores are not an inert medium; many chemical and physical processes take place in the ice. Under certain conditions, and for certain compounds, these processes can alter the signal stored in the enclosed air bubbles over time. The measured signals then no longer correspond exactly to the past composition of the atmosphere. Production of CO2 in Greenland ice cores and N2O in both Greenland and Antarctic ice cores are prominent examples for such an alteration of the atmospheric signal (Stauffer et al. 2003).
N2O: A growing climate threat
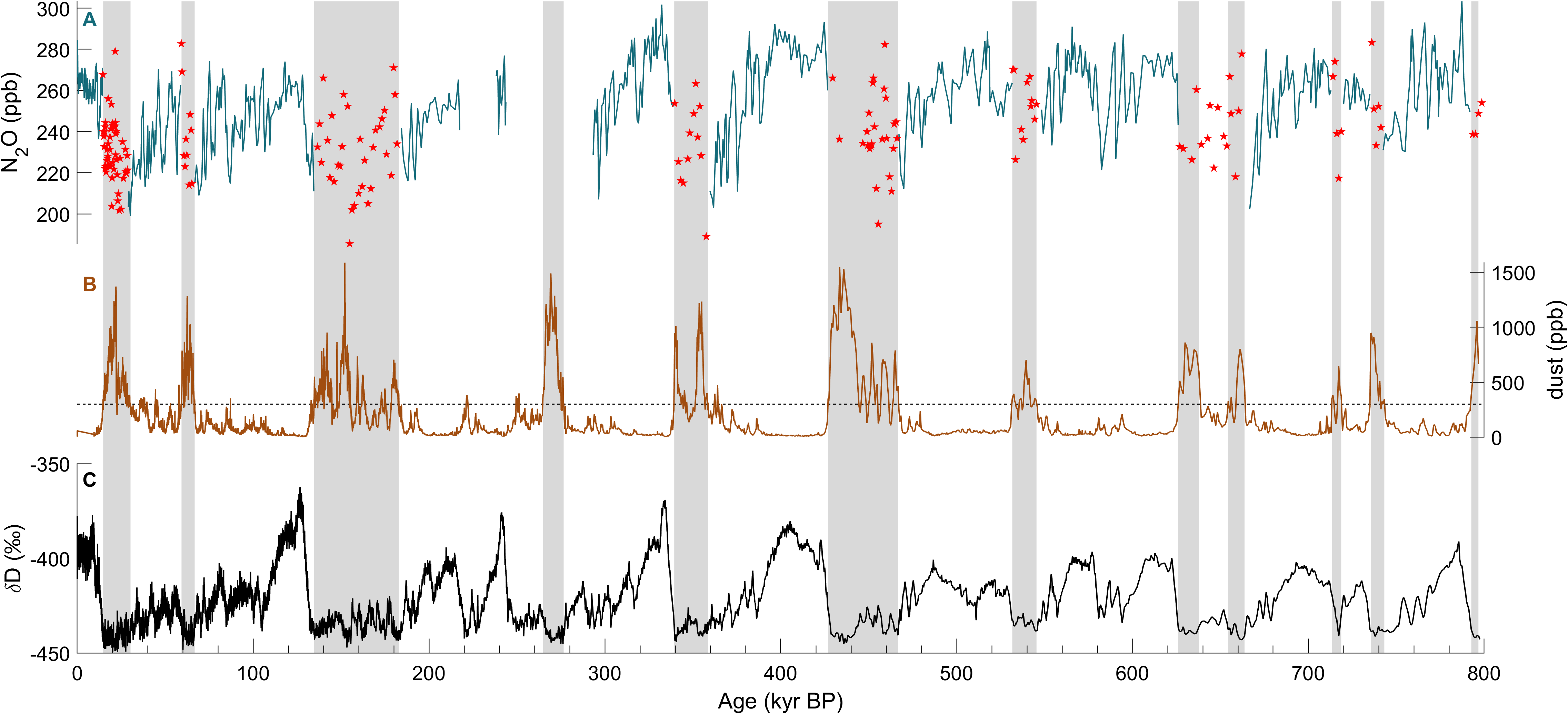
The study of N2O is important as it is a potent greenhouse gas that is also involved in the destruction of stratospheric ozone. The atmospheric concentration of N2O, with a global warming potential 273 times higher than CO2, has been increasing continuously over the past 150 years, reaching 332 ppb in 2019 (IPCC 2021). Currently, anthropogenic (mainly agricultural) sources contribute 43% of total N2O emissions, and natural sources from soils and oceans account for 57% (Tian et al. 2020). The main N2O sink is photochemical destruction in the stratosphere, and its preindustrial atmospheric lifetime is 123 years (Prather et al. 2015). Warmer climate seems to enhance natural N2O emissions, resulting in a positive feedback (Schilt et al. 2010a). This effect is difficult to predict because present and past N2O dynamics are poorly understood (Fischer et al. 2019). Reconstructed atmospheric N2O concentrations vary substantially on glacial-interglacial timescales (Flückiger et al. 2004; Schilt et al. 2010a). However, significant parts of the 800-kyr atmospheric record of N2O are missing due to in situ formation of N2O in glacial ice, rich in mineral dust (Fig. 1).
In situ production of N2O
Several observations indicate that the N2O concentrations measured in the ice are affected by a non-atmospheric source. For example, ice cores from different drilling sites show significantly different N2O values for given time periods (Schilt et al. 2010a, b). Considering the long atmospheric lifetime of N2O and, as a result, its geographically homogenous atmospheric concentration, this observation is inconsistent with only atmospheric N2O variations. The non-atmospheric source alters the N2O records exclusively during glacial periods, when the dust concentrations are high (Fig. 1). This indicates a production of N2O from compounds in, or attached to, aeolian dust deposited onto the ice sheet (Schilt et al. 2010a).
For most Antarctic ice cores, the dust-rich sections are almost entirely affected by in situ N2O production (Schilt et al. 2010a). Comparing different ice cores from Antarctica, the highest N2O concentrations are found in ice cores with the highest dust levels (Schilt et al. 2010b). In contrast, in situ N2O production in Greenland ice is not correlated with dust concentrations, and mainly occurs at the beginning and end of the Dansgaard–Oeschger events (Flückiger et al. 2004). Because these climatic transitions are associated with changes in chemical composition of the dust, N2O production is likely controlled by this factor.
Previous approaches
In situ N2O production represents a challenge for reconstructing the past atmospheric concentrations of N2O during glacial periods. To avoid misinterpretation in terms of past climatic variations and derived changes in marine and terrestrial sources, in situ production must be systematically detected.
Flückiger et al. (2004) used a detection algorithm for Greenland ice that iteratively identifies N2O values exceeding a threshold of 8 ppb, which is about 3% of the typical glacial atmospheric concentrations, above a smoothing spline calculated through the whole dataset. However, this algorithm is only applicable to high-resolution datasets affected by erratic outliers, and is not valid for sharp rises in atmospheric concentration that could be mistaken for N2O outliers. The second approach, applied to lower-resolution records from Antarctic ice cores, identifies samples with a dust concentration above 300 ppb (Fig. 1; Spahni et al. 2005). This dust threshold is purely empirical and does not reflect the complexity of the N2O production. Indeed, if in situ N2O production in Antarctic ice is roughly proportional to the dust content, samples below this threshold may still be affected (e.g. at 640 kyr BP in Fig. 1). In summary, such detection algorithms help to improve the records, but are heuristic at best, and do not allow us to correct for the in situ contribution. For this, process understanding of the in situ formation is required.
A new process-based quantification method
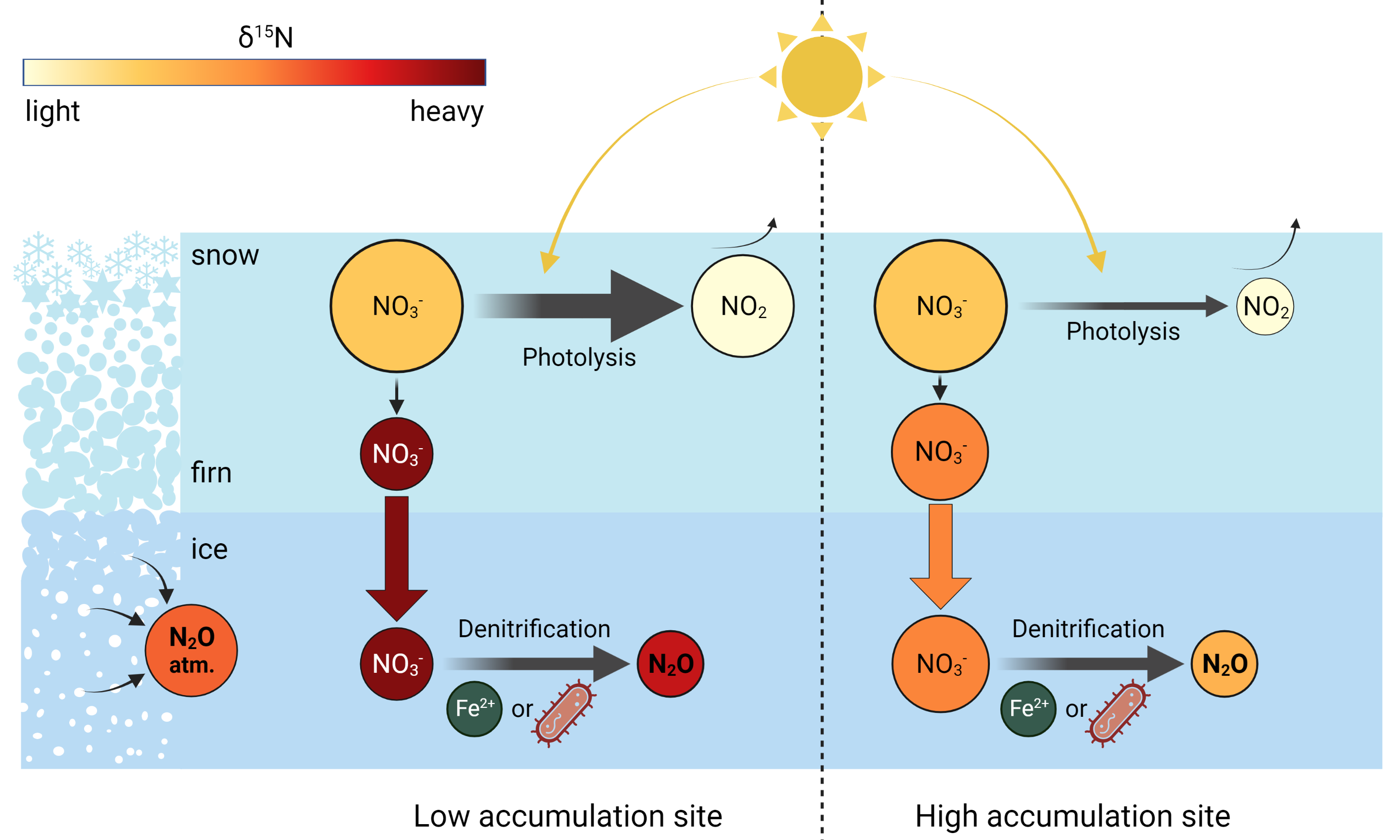
In the framework of the DEEPICE project, we seek to understand the processes responsible for N2O production. By identifying the chemical reaction at play, and the specific conditions necessary for its occurrence, we target three objectives: 1) to systematically detect the samples affected by in situ N2O to avoid misinterpretation of atmospheric N2O variations; 2) to quantify and predict the amount of in situ production in ice samples; and 3) to correct the N2O measurements to isolate the atmospheric N2O signal. Since isotope analysis is a powerful tool to trace sources of a compound, and previous work showed that the in situ fraction has an isotopic signature distinct from the atmospheric fraction (Fischer et al. 2019; Sowers 2001), our approach is based on the isotope analysis of N2O and impurities that may be precursors of the N2O in situ produced (Fig. 2).
Investigating the reaction consists, first of all, in identifying the precursors and reactants. The two main production pathways for N2O are nitrification, i.e. conversion of ammonium (NH4+) to nitrate (NO3-) with N2O as a byproduct, and denitrification, i.e. reduction of NO3- to N2O (Baggs 2011). The amounts of NO3- and NH4+ in ice are both more than enough to form the observed amounts of in situ N2O. Comparing N2O data from different ice cores, we observe that in drilling sites with low snow accumulation rates the in situ fraction of N2O has high δ15N values compared to the atmospheric fraction. This is the case in the Vostok (Sowers 2001) and EDC (unpublished data) ice cores. The δ15N value of NO3-, impacted by post-depositional processes, is also higher with a decreasing accumulation rate. Indeed, NO3- photolysis in snow is accentuated in low accumulation sites, inducing high enrichment in 15N (Fig. 2; Erbland et al. 2013). These similar isotopic enrichments make NO3- a good candidate as a precursor of in situ N2O. To test this hypothesis, we are currently performing joint measurements of the nitrogen and oxygen isotopic compositions of N2O and NO3- in the same samples. The in situ fraction of N2O and its isotopic composition are calculated using a mass balance approach, with atmospheric values as defined by the almost unaffected 140-kyr record from the Talos Dome ice core (Schilt et al. 2010b). Correlated isotopic signatures of NO3- and in situ N2O would point to a denitrification reaction.
Our next step is to take a closer look at the production pathway. Denitrification can be performed by bacteria and through abiotic processes (Fig. 2). For example, NO3- is reduced to N2O by Fe2+ (Samarkin et al. 2010). As the intramolecular distribution of 15N in the N2O molecule (15N-14N-O or 14N-15N-O) only depends on the production pathway, we plan to measure the position-dependent isotope ratio of nitrogen in N2O to distinguish between a biotic or abiotic reaction.
Conclusion
A precise understanding of the processes leading to N2O in situ production represents a major step forward in interpreting and completing the N2O record, which would complement the existing Antarctic CO2 and CH4 records over the last 800 kyr. Knowing the past variations of the three most important greenhouse gases is crucial to understanding the climate system, and, thus, better predict future climate change.
affiliation
1Climate and Environmental Physics, Physics Institute and Oeschger Centre for Climate Change Research, University of Bern, Switzerland
contact
Lison Soussaintjean: lison.soussaintjean@unibe.ch
references
Baggs EM (2011) Curr Opin Environ Sustain 3: 321-327
Erbland J et al. (2013) Atmospheric Chem Phys 13: 6403-6419
Fischer H et al. (2019) Biogeosciences 16: 3997-4021
Flückiger J et al. (2004) Glob Biogeochem Cycles 18: GB1020
IPCC (2021) Climate Change 2021: The Physical Science Basis. Cambridge University Press, 2391 pp
Lambert F et al. (2008) Nature 452: 616-619
Landais A, Stenni B (2021) Water stable isotopes (δD, δ18O) data from EDC ice core. PANGAEA
Prather MJ et al. (2015) J Geophys Res Atmos 120: 5693-5705
Samarkin VA et al. (2010) Nat Geosci 3: 341-344
Schilt A et al. (2010a) Quat Sci Rev 29: 182-192
Schilt A et al. (2010b) Earth Planet Sci Lett 300: 33-43
Sowers T (2001) J Geophys Res Atmos 106: 31903-31914
Spahni R et al. (2005) Science 310: 1317-1321