- Home
- Publications
- PAGES Magazine
- Challenges of Water Isotope Measurements On Ice Cores
Challenges of water isotope measurements on ice cores
Eirini Malegiannaki, K.M. Peensoo, P. Bohleber and V. Gkinis
Past Global Changes Magazine
31(2)
64-65
2023
Due to thinning of ice-core layers, the deepest part of the cores is the hardest to analyze. New micro-destructive and high-resolution instrumental approaches are necessary to retrieve a water-isotope record that accurately represents past climate variations.
Water isotopic composition of ice cores
When water travels from the evaporation to the precipitation site, the different water isotopes (H216O, H218O, HD16O) modify its isotopic signature, represented by δ18O and δD. For example, a lighter isotope, such as H216O, evaporates more easily than a heavy one, such as H218O, leading to a temperature-dependent fractionation and the establishment of a relationship between water isotopic composition and condensation temperature (Dansgaard 1953).
Ice cores exhibit distinctive layered characteristics where the water-isotopic signature is preserved. Due to the preservation of ice for long time periods in large ice sheets, water-isotope data offers access to a continuous and high-resolution record of past climatic variability. This is shown in the climate records generated during the EPICA (1996–2008) deep-core project which extend up to 800,000 years ago. The ongoing Beyond EPICA (2019–2026) project aims to extend the ice-core record up to 1.5 Myr in order to reconstruct Antarctica’s climate history.
However, analyzing deep ice cores poses challenges. Thinning of annual layers in the deeper sections of an ice sheet, and post-depositional processes within the ice core, such as water molecule diffusion within the ice, may result in the smoothing of the water-isotope signal (Johnsen et al. 2000). This challenges the recovery of the original isotopic signal of the deposited precipitation. In addition, there are limitations in the measurement procedures which further increase difficulty in retrieving the original climate signal.
Analytical techniques for water-isotope measurements on ice cores
Advances in sample preparation and signal acquisition techniques have significantly enhanced the capability of obtaining high-resolution water-isotopic data from ice cores. Isotope Ratio Mass Spectrometry (IRMS) is an analytical technique traditionally employed to acquire water-isotope signals that offer high-precision measurements. However, due to the “sticky” nature of the water molecule, and its condensable gas phase, the water should be converted into a chemical form that is compatible with the IRMS, which is a time-consuming process and prone to inaccuracies. Laser-based water-isotope measurement techniques display the potential to tackle the aforementioned issues (Kerstel and Gianfrani 2008), as well as other difficulties related to the expensive and bulky instrumental equipment needed.
Cavity Ring Down Spectroscopy (CRDS) is a sensitive spectroscopic technique that uses laser light and an optical cavity to measure water-isotope ratios. For the measurement, a laser pulse is injected into an optical cavity, and the decay time, or "ring down" time, of the detected light intensity is measured. This decay time is affected by the presence of gas molecules in the cavity, indicating the gas concentration. By coupling specific laser wavelengths with absorption features of the target molecules, CRDS allows for highly sensitive measurements.
CRDS has emerged as a preferred alternative to IRMS, also offering the ability to perform field measurements. Different ice-sample preparation methods enable water-isotope measurements using a CRDS analyzer in either continuous or discrete mode, respectively. However, it is important to note that both approaches require substantial time and sample consumption.
Laser Ablation: a fast micro-destructive sampling method
Laser-matter interaction has been widely used for developing new techniques, including micromachining, laser cutting, sampling, and material development. When a pulsed laser beam is focused on a target surface, a high amount of energy is confined in space and time in the irradiated area, which can cause material ejection in the gas phase, known as Laser Ablation (LA). Ice sampling using LA serves a dual purpose: 1) It provides high-resolution analysis, and 2) it uses the smallest amount of sample possible for analysis, given the micro-destructive nature of the ablation process. In this way, the ice core is not melted and the same sample can be used for other types of analysis while, at the same time, it is possible to obtain high-quality continuous measurements.
LA has already been adopted as a sampling method in elemental concentration studies on ice cores (Della Lunga et al. 2017). Recently, LA sampling allowed for fast two-dimensional imaging of the impurities in ice at very high resolution (just a few tens of micrometers), generating new insights into ice-impurity interactions (Bohleber et al. 2021).
Laser Ablation–Cavity Ring Down Spectroscopy on ice cores
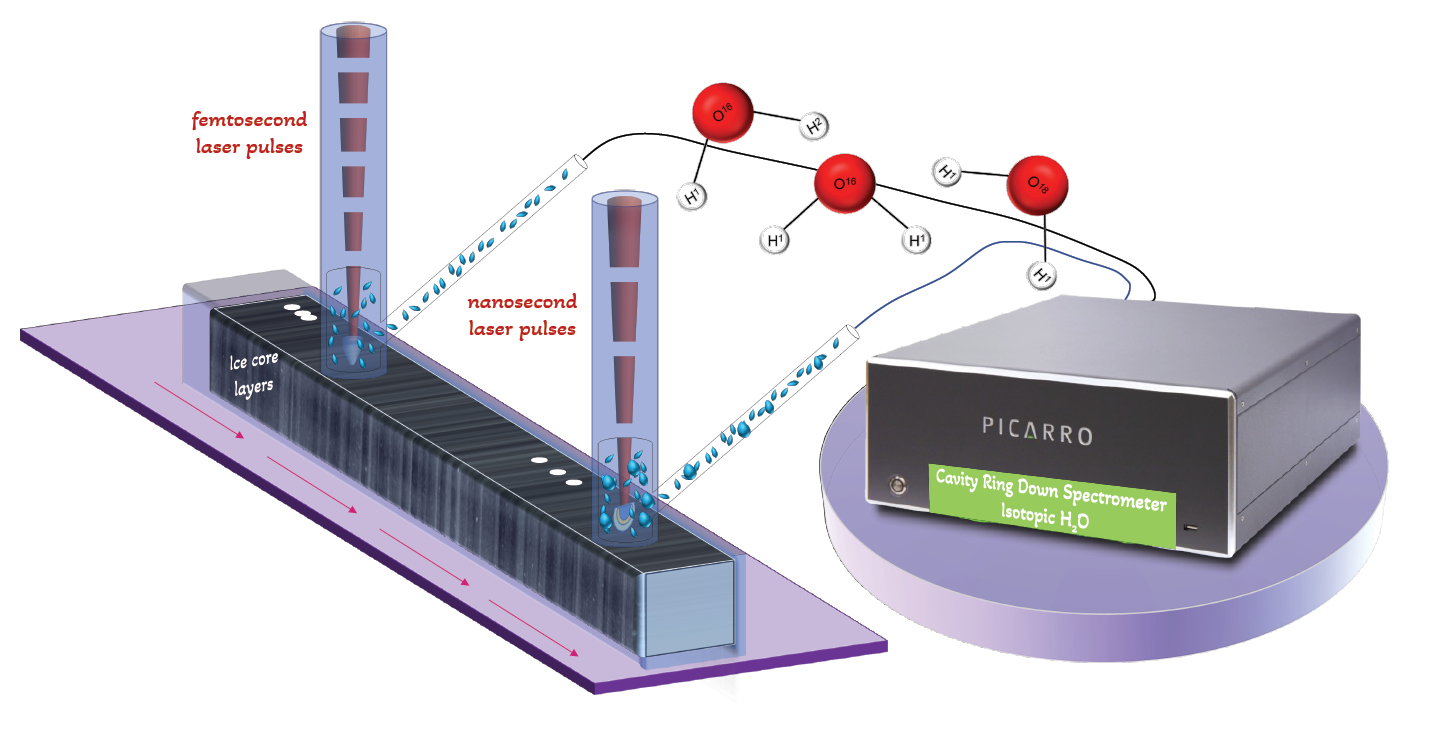
Laser Ablation (LA) and Cavity Ring Down Spectroscopy (CRDS) techniques have recently been used together to enable high-quality water-isotopic measurements on ice cores. The conceptual design of the LA-CRDS system is illustrated in Figure 1. The experimental setup for LA sampling on ice includes essential components such as a focused laser beam, a motorized translational stage for accurate ice movement, an ablation chamber to collect the sampled material, and a transfer line that transports it to the CRDS analyzer. These components work together to enable efficient and reliable analysis of ice samples in a continuous manner.
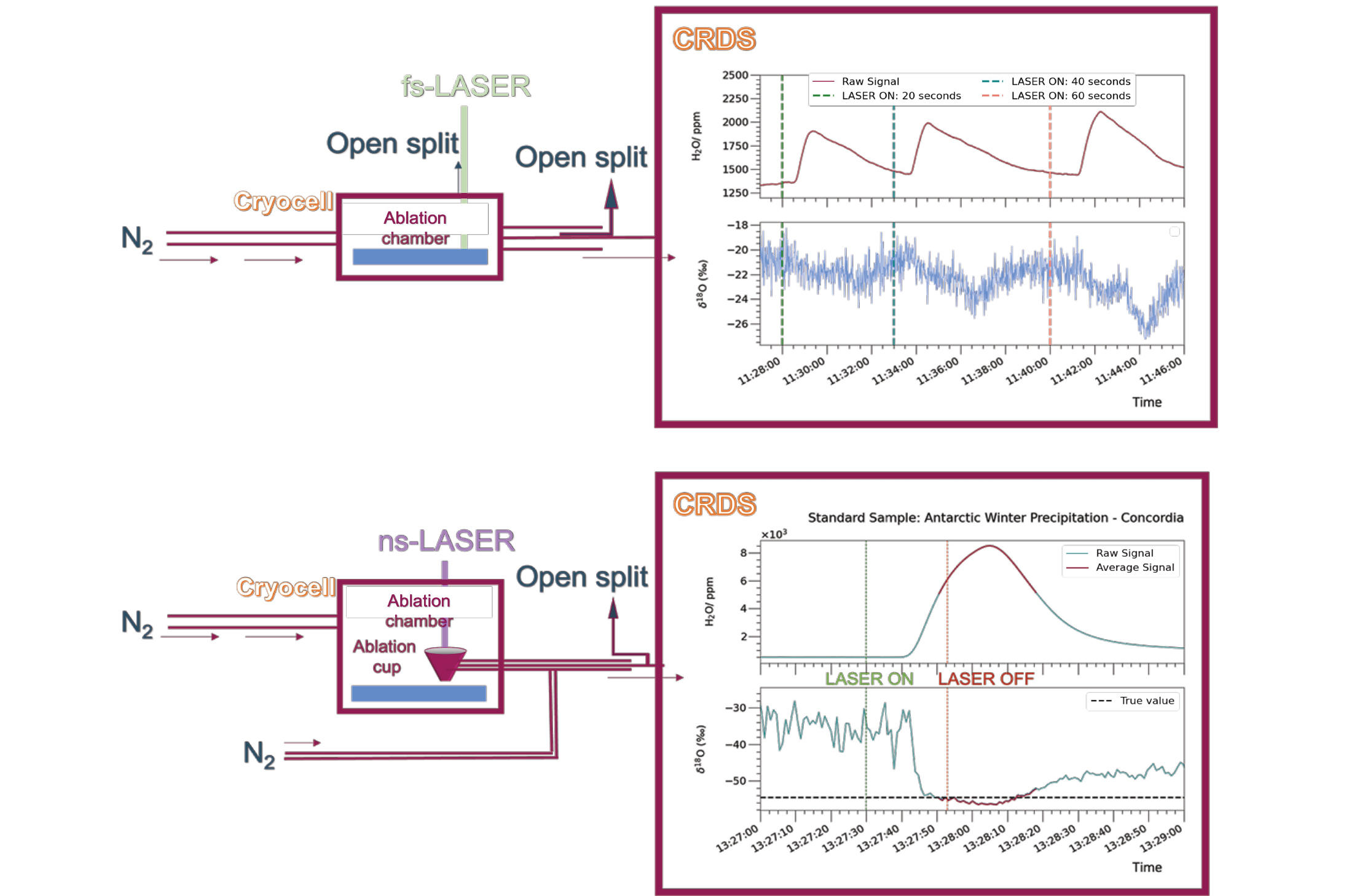
During sampling, the laser interacts with the ice and creates a mixture of vapor and tiny particles. The laser can produce short pulses, either in the nanosecond or femtosecond range. The amount of time the laser interacts with the ice affects the composition of the mixture. One challenge in measuring water isotopes is that when the ice melts or undergoes incomplete phase changes, it can change the isotopic signature of the material being studied. However, using very short laser pulses in the femtosecond range can help reduce this issue, because the interaction time is shorter than the time it takes for heat to spread in the material.
The compatibility between the vapor produced by the LA sampling process and the gas analyzer, CRDS, led to the inception of the LA-CRDS technique. This innovative design enables fast gas-phase sample collection directly from the ice, facilitating high-quality water-isotopic measurements. The successful implementation of this approach in experimental laboratories has yielded promising results.
Both femtosecond (Fig. 2a; Kerttu 2021) and nanosecond laser pulses (Fig. 2b) have been applied in two distinct experimental configurations coupled with a CRDS analyzer, leading to the detection of the ablated ice (artificial ice samples produced from purified and liquid water from Antarctic winter precipitation) and acquisition of the respective water-isotope signal. These initial results demonstrate the feasibility of ablating ice material with lasers of different pulse durations, and acquiring a water-isotope signal using the CRDS analyzer.
Conclusions
Advances in water-isotope measurement techniques, both on the sampling and the detection side, had already shown a big impact on the retrieval of continuous high-resolution water-isotope records from ice cores. By combining precise analyzers with melting methods, scientists can obtain detailed data.
The introduction of LA, coupled with CRDS analyzers, presents a promising approach for capturing high-resolution isotope signals, especially from the deepest parts of the ice cores. This micro-destructive technique minimizes sample usage while providing valuable insights into laser-ice interaction principles for accurate interpretation of the water-isotope instrumental signal. The synergistic work on diffusion studies (Gkinis et al. 2014; Holme et al. 2018), and the development of high-quality water-isotope measurements, will further enhance the precision of past-temperature estimations.
affiliationS
1Physics of Ice, Climate, and Earth, Niels Bohr Institute, University of Copenhagen, Denmark
2Department of Environmental Sciences, Informatics, and Statistics, Ca’Foscari University of Venice, Italy
contact
Eirini Malegiannaki: eirini.malegiannaki@nbi.ku.dk
references
Bohleber P et al. (2021) The Cryosphere 15: 3523-3538
Dansgaard W (1953) Tellus 5: 461-469
Della Lunga D et al. (2017) The Cryosphere 11: 1297-1309
Gkinis V et al. (2014) Earth Planet Sci Lett 405: 132-141
Holme C et al. (2018) Geochim Cosmochim Acta 225: 128-145
Johnsen S et al. (2000) Phys Ice Core Rec 121-140
Kerstel E, Gianfrani L (2008) Appl Phys B Lasers Opt 92: 439-449
Kerttu MP (2021) MSc Thesis, University of Copenhagen, 46 pp